Assistant Professor Claudia Fallini hopes the research will lead to new Alzheimer’s disease insights and improved stroke outcomes.
Continue reading "$413,729 NIH grant helps to study the link between stroke and Alzheimer’s disease"Category: News
Van Nostrand lab collaborates with Eli Lilly to investigate brain bleed risk in Alzheimer’s disease treatments
The project will help shed light on whether there may be a way to reduce or eliminate the risk.
Continue reading "Van Nostrand lab collaborates with Eli Lilly to investigate brain bleed risk in Alzheimer’s disease treatments"$8.5 million in NIH grants support Ryan Institute research to help treat cerebral palsy
Assistant Professor Katharina Quinlan is principal investigator on two new grants that could repurpose existing therapeutics to treat the disorder.
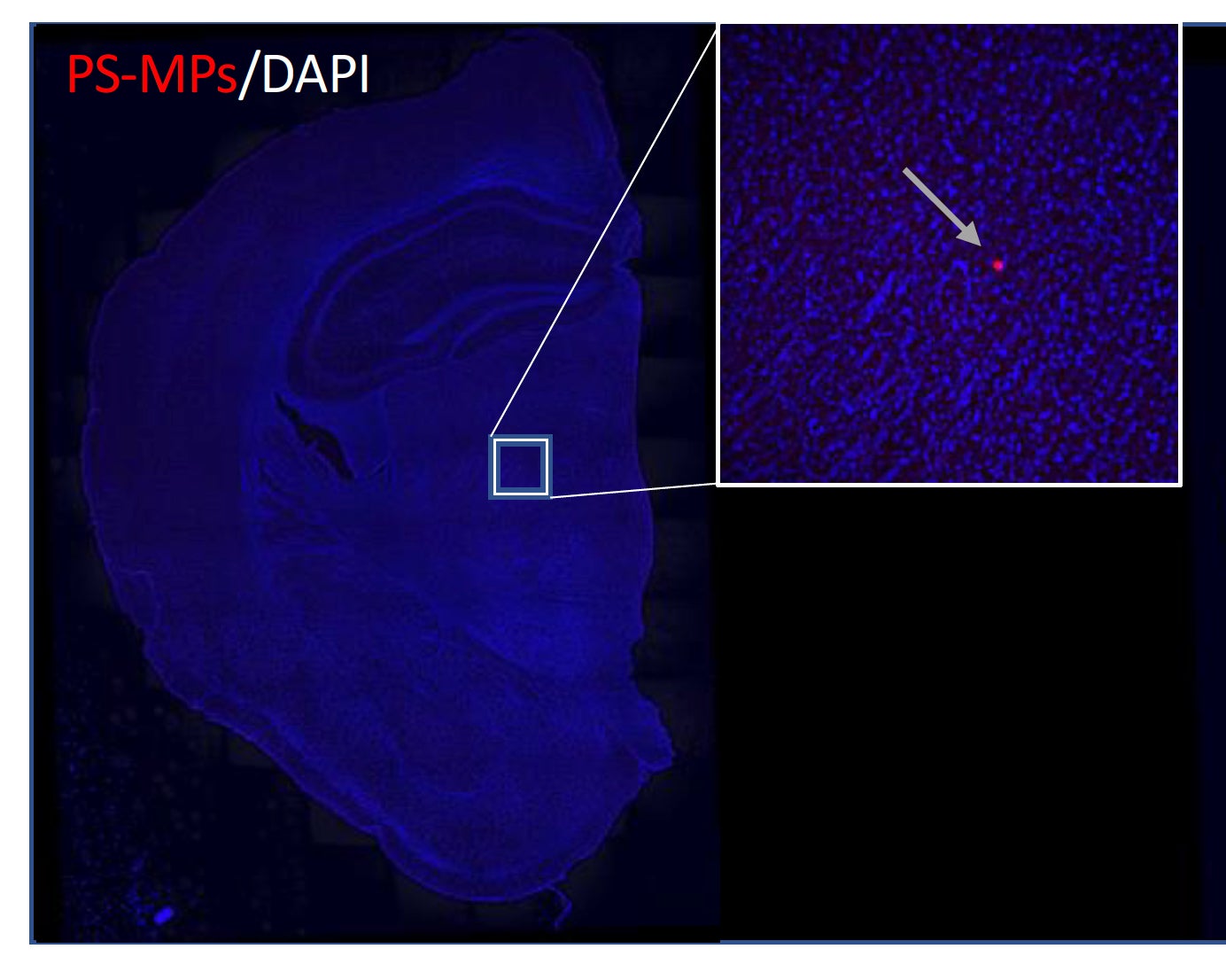
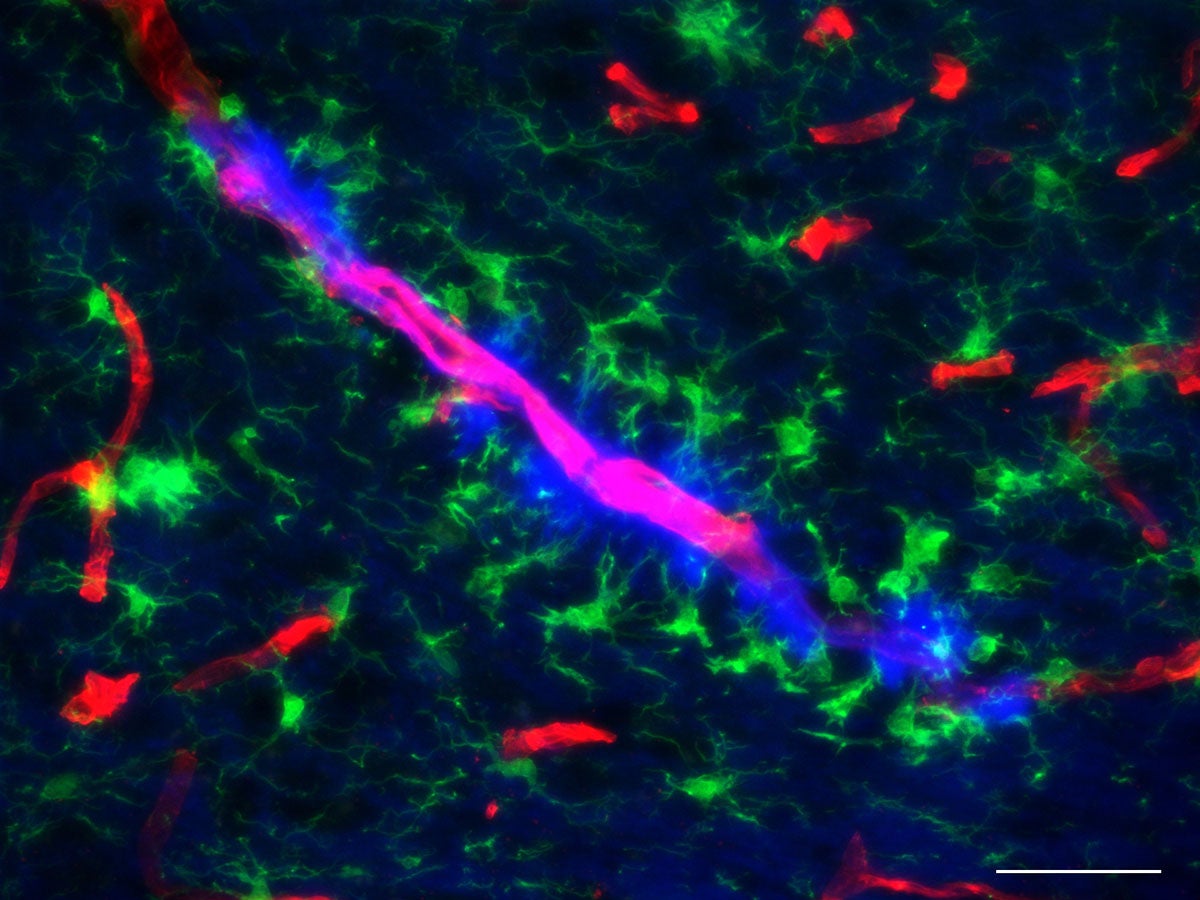
Continue reading "$8.5 million in NIH grants support Ryan Institute research to help treat cerebral palsy"A new study investigates the impact of microplastics in the brain
Assistant Professor Jaime Ross and her lab report surprising findings on the impact of short-term microplastics exposure through drinking water.
Continue reading "A new study investigates the impact of microplastics in the brain"Van Nostrand lab is part of $8 million grant for U.S. and European consortium on brain clearance in CAA
William Van Nostrand, co-executive director of the George & Anne Ryan Institute for Neuroscience at the Univeristy of Rhode Island, is part of a team awarded a five-year, $8 million grant from the prestigious Leducq Foundation.
Continue reading "Van Nostrand lab is part of $8 million grant for U.S. and European consortium on brain clearance in CAA"New grant from Cure Alzheimer’s Fund supports research into the link between ApoE4 and CAA
A new grant supports research by the Van Nostrand lab to investigate the effects of the ApoE 4 genotype in cerebral amyloid angiopathy, a disorder that commonly occurs with Alzheimer’s disease.
Continue reading "New grant from Cure Alzheimer’s Fund supports research into the link between ApoE4 and CAA"Ryan Institute expert discusses new Alzheimer’s disease research showing that changes in the brain also can be seen in the eyes
Assistant Professor Jess Alber answers questions about new research that supports her own work toward screening for Alzheimer’s disease during routine eye exams.
Continue reading "Ryan Institute expert discusses new Alzheimer’s disease research showing that changes in the brain also can be seen in the eyes"First-in-kind models will help break new ground in studying Alzheimer’s disease and related disorders
Gene-edited rat models will reflect vascular pathology associated with Alzheimer’s and related disorders.
Continue reading "First-in-kind models will help break new ground in studying Alzheimer’s disease and related disorders"Ryan Institute researchers are part of breakthrough studies on “mature” neurons, epigenetics in aging
Three Ryan Institute faculty are co-authors on breakthrough studies at Northwestern University and Harvard Medical School.
Continue reading "Ryan Institute researchers are part of breakthrough studies on “mature” neurons, epigenetics in aging"Ryan Institute Co-Director John Robinson named Thomas M. Ryan Professor of Neuroscience
The URI Foundation & Alumni Engagement (URIFAE) is pleased to announce that George and Anne Ryan Institute for Neuroscience Co-Executive Director John K. Robinson has been named Thomas M. Ryan Professor of Neuroscience.
Continue reading "Ryan Institute Co-Director John Robinson named Thomas M. Ryan Professor of Neuroscience"